Make yourself Familiar with the instrument (hover for more info)
 Measuring cylinder
Measuring cylinder
 Conical Flask
Conical Flask
 Weighing Scale
Weighing Scale
 Burner
Burner
 Burette
Burette
Step 1: Select the sample




Step 1-1: Melt the solid Mixture
 click to turn on hot
plate
click to turn on hot
plate
 click to place
flask
click to place
flask
Generally fats are solid at room temperature
Step 2: Weigh 5g of oil/fat sample in conical flask
 click to place flask
click to place flask

What is the required quantity of sample to be taken for estimation of peroxide value of oil/fat
Step 3: Add 30 ml acetic acid-chloroform solution in the sample flask and swirl to dissolve
 click to pour
acid solution
click to pour
acid solution


In what ratio acetic acid and Chloroform solution is prepared
Step 4: Add 0.5 ml saturated KI solution and shake it for 1 min


 click to suck KI
solution
click to suck KI
solution
Step 5: Add 30 ml distilled water
 click to pour
distilled water
click to pour
distilled water
 click to pour
distilled water
click to pour
distilled water


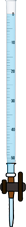
Step 6: Titrate the mixture with 0.1 N sodium thio-sulphate solution until yellow color is almost gone

0 ml
 click to pour
0.1N NaOH
click to pour
0.1N NaOH
Step 7: Add 0.5 ml 1% starch solution




 click to suck
solution
click to suck
solution
Which indicator is used in titration for estimation of peroxide value of oil/fat
Step 8: Further titrate until blue color just disappears


0 ml
Observation Table
| Selected sample | Weight of sample (g) (W) | Normality of Sodium thiosulphate solution (N) | Burette reading (ml) | Titrant used (ml) | |
|---|---|---|---|---|---|
| Initial value | Final value | ||||
| 5.0 | 0.1 | 0.0 | |||
FFA(%) =

| Peroxide value (mEq peroxide/kg) = | V x N |
|---|---|
| W | |
| Peroxide value (mEq peroxide/kg) = | x 0.1 |
| 5 | |
| Peroxide value (mEq peroxide/kg) = |
Fresh oil sample has less peroxide value than used oil which indicates its good quality and better shelf-life
Inference
The rancid oil has the peroxide value (mEq peroxide/kg) in the range of
The rancid oil has the Peroxe value in the range of 30-40 mEq/kg as per FSSAI standard.
The peroxide value of the given sample is 9.2%. So, this sample is safe for edible purposes.